Which Kind of Force Is a Dipole-dipole Force Apex
This is an intermolecular force. Although molecules in a liquid are in constant motion they tend to align in the lowest energy orientation which would be to maximize attractions parts a b of fig.

Intermolecular Force Chemistry Organic Chemistry
YOU MIGHT ALSO LIKE.
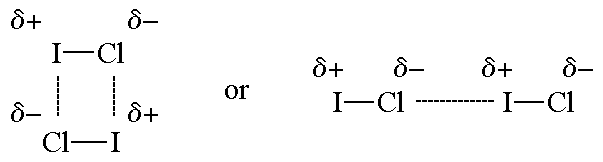
. Intermolecular Forces Select the choice that best answers the question or completes the sentence then ENCIRCLE the letter. Dipole-dipole forces are attractive forces between molecules that possess permanent dipoles. Unit 4- Types of Bonding Learn with flashcards games and more for free.
Three types of intermolecular forces exist between electrically neutral molecules. Up to 24 cash back This type of bonding only exists between polar molecules. Dipole-dipole forces have strengths that range from 5 kJ to 20 kJ per mole.
1131 and minimize repulsion parts c d of fig. Dipole-Dipole Force c London Dispersion Force. Dipoledipole forces occur between molecules with permanent dipoles ie polar molecules.
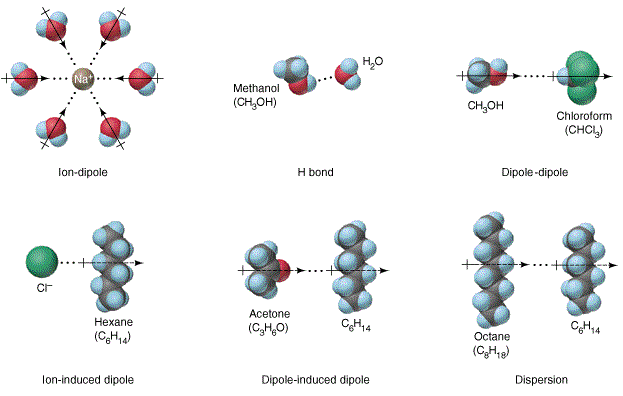
And another kind of attractive force ion-dipole is important in solutions. Having a permanent nature dipole-dipole forces are stronger than dispersion forces. Dipole-dipole forces are stronger than dispersion forces as long as the molecules being compared have approximately the same mass.
Now we know that since the dipole is having positive and negative charge separated by some distance so here when one dipole is. For molecules of similar size and mass the strength of these fo. Related Threads on Dipole-Dipole Force Why NCl3 is dipole-dipole in intermolecular force.
These are intermolecular forces. The atoms share the electrons unequally because the more electronegative atom pulls the shared electrons toward itself. Der Waals forces 9a Which kind of force is a dipole-dipole force.
Dipole-dipole forces are the attraction between the positive end of one molecule and the negative end of another. 21 KB Views. Keesom forces Debeye forces London forces And H-bonding What type of forces are dispersion forces dipole-dipole forces and hydrogen bonds.
Dipole-dipole forces are attractive forces between the positive end of one polar molecule and the negative end of another polar molecule. Helpful 0 Not Helpful 1 Add a Comment. The δ- end of one polar molecule is attracted to the δ end of another drawing them together.
Dipole-dipole forces are intermolecular forces that occur between polar molecules. Attractive forces between the positive end of one polar molecule and the negative end of another polar molecule. Post reply Insert quotes Share.
When molecules have permanent dipole moment then two molecules will apply force on each by interacting with the electric field of each other. Dipoles form when there is a large difference in electronegativity between two atoms joined by a covalent bond. Where dipole dipole forces mostly occur.
Answers and Replies LaTeX Guide BBcode Guide. The attraction between the oppositley charged regions of the molecule is called Dipole-Dipole bond. London dispersion forces dipole-dipole attractions and hydrogen bonding.
Remember that an ion is an atom that has gained or lost one or more electrons and therefore has. These are electrostatic forces. Waters dipole moment has a positive center between the two nuclei of the hydrogens.
It is a force between an ion and a dipole molecule. Within a polar molecule there is a partially positive region and a partially negative region. These are the polar molecules.
Choice Intermolecular attraction Intramolecular dipole formation Intramolecular bond formation. When forming this type of force the polar molecules tend to be aligned so that the attraction between the molecules. They are much weaker than ionic or covalent bonds and have a significant effect only when the molecules involved are close together touching or almost touching.
An ion-dipole force is just what its name says. As the name implies dipole-dipole forces are those between dipoles when they are very close together or touching one another. As we know that electric field due to dipole on its axial position is given as.

The Relative Strength Of Hydrogen Bonding Dipole Dipole Interactions And London Dispersion Forces Organic Chemistry Chemistry Teaching Game

Chemistry Infographic Intermolecular Forces Video Chemistry Lessons Chemistry Intermolecular Force
No comments for "Which Kind of Force Is a Dipole-dipole Force Apex"
Post a Comment